Here are important questions and answers of carbon and its compounds class 10
1 mark Questions:
Ques 1. What is tetravalency?
Ans. It is the ability to form four chemical bonds.
Ques 2. why is carbon able to form multiple bonds?
Ans. Carbon is able to form multiple bonds because it is tetravalent in nature which allows it to make single, double or even triple bonds to attain a stable electronic configuration.
Ques 3. What is isomerism?
Ans. When two or more compounds have same molecular formula but different structures, they are said to be ‘isomers’. And this phenomenon is known as Isomerism.
Ques 4. Define the property of catenation of carbon.
Ans. Catenation is the self linking property of carbon to form long or branched chains
Ques 5. What is a functional group? Give an example.
Ans. A functional group is a substituent in a molecule that is responsible for its characteristic chemical properties. eg. Alcohol (-OH)
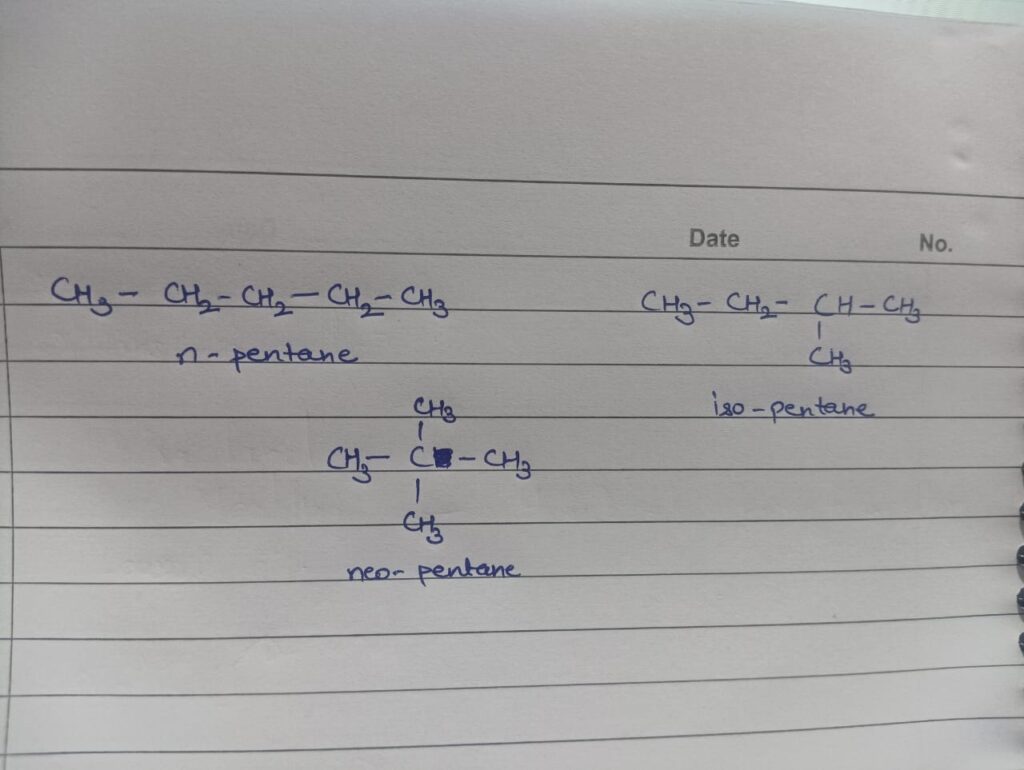
Ques 6. Give the chain isomers of pentane.
Ans. Pentane has three isomers:
Ques 7. What is a homologus series?
Ans. Basically, it is a group of organic compounds having the same functional group and similar chemical properties.
Ques 8. What are alkenes?
Ans. Alkenes are the unsaturated hydrocarbons (having carbon-carbon double bond) with the general formula CnH2n.
Ques 9. Why boiling point of alcohols are generally higher than their corresponding alkanes?
Ans. Because of the presence of strong hydrogen bonding in alcohols (-OH), they have higher melting and boiling points.
Ques 10. Why is the melting and boiling point of C4H8 higher than that of C3H6 and C2H4?
Ans. These are the members of homologous series and its each member has 1 carbon and 2 hydrogen atoms more than the previous member. Due to which its molecular mass increases and hence the melting and boiling point.
Ques 11. Why do we not see any gradation in the chemical properties of homologous series of compounds?
Ans. It is the functional group that determines the chemical properties, and all members of a homologous series share the same functional group.So, we can’t see any gradation in their chemical properties.
Ques 12. Write the molecular formula of any two consecutive members of homologous series of aldehydes.
Ans. Methanal H-CHO (CH2O) and Ethanal CH3-CHO (C2H4O).
Ques 13. Carbon compounds have low melting and boiling points. why?
Ans. This is because weak van der Waals forces hold the bonds in carbon compounds, making them easy to break.
Ques 14. Explain why carbon compounds don’t conduct electricity.
Ans.To conduct electricity, free electrons are required, but carbon compounds cannot do so because they form covalent bonds by sharing electrons.
2 marks Questions:
Ques 15. Give reason why carbon can neither form C4+ cations nor C4- anions but form covalent bonds.
Ans. Carbon can not form C4+ cation because it takes a lot of energy to lose 4 electrons and also it can’t form C4- anion because it would be difficult for the nucleus with six protons to hold on to ten electrons.
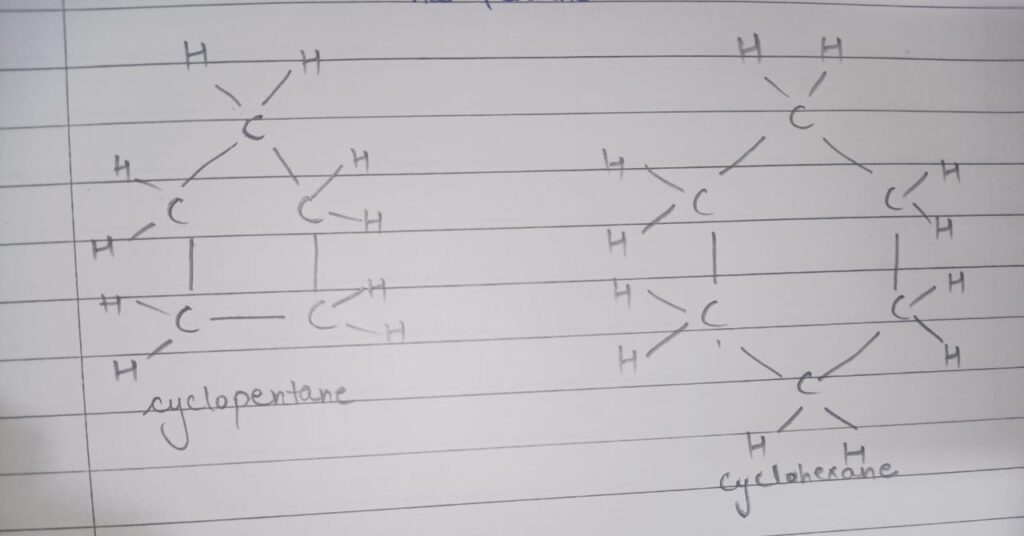
Ques 16. Name two cyclic hydrocarbons and draw the structure of each.
Ans. cyclopentane and cyclohexane
Ques 17. List three differences between covalent and ionic compounds.
Ans.
| Covalent compound | Ionic compound |
| 1. Covalent compounds are formed by sharing of electrons. | 1. Ionic compounds are formed by gaining or losing of electrons. |
| 2. They have generally low melting and boiling point. | 2. They have generally high melting and boiling point. |
| 3.The bonds are generally formed between non metals. | 3.The bonds are generally formed between metal and non metal. |
| Eg. H2O | Eg. NaCl |
Ques 18. Distinguish between a saturated and unsaturated hydrocarbon by flame test. List the products of combustion reaction of saturated hydrocarbon.
Ans. Saturated hydrocarbons generally give a clean flame while unsaturated carbon compounds give a yellow sooty flame with lots of black smoke.
Ques 19. Name two oxidising agents used for the conversion of alchols to acid.
Ans. Potasium permanganate(KMnO4) and potassium dichromate (K2Cr2O7).
Ques 20. Write physical properties of carbon compounds.
Ans. 1. They have low melting and boiling points.
2. They do not conduct electricity.
3. They are generally insoluble in water.
Ques 21. Write the name and structures of (i) aldehydes and (ii) ketone with molecular formula C3H6O.
Ans. Aldehyde- CH3-CH2-CHO and Ketone- CH3-CO-CH3
Ques 22. Write the properties of homologous series.
Ans. 1. All members of homologous series have the same general formula.
2. The two consecutive members of series are differ by -CH2 group and 14amu.
3. There is no gradation in their chemical properties.
Ques 23. Draw the structure of benzene and hence write the number of single and double bonds in it.
Ans.
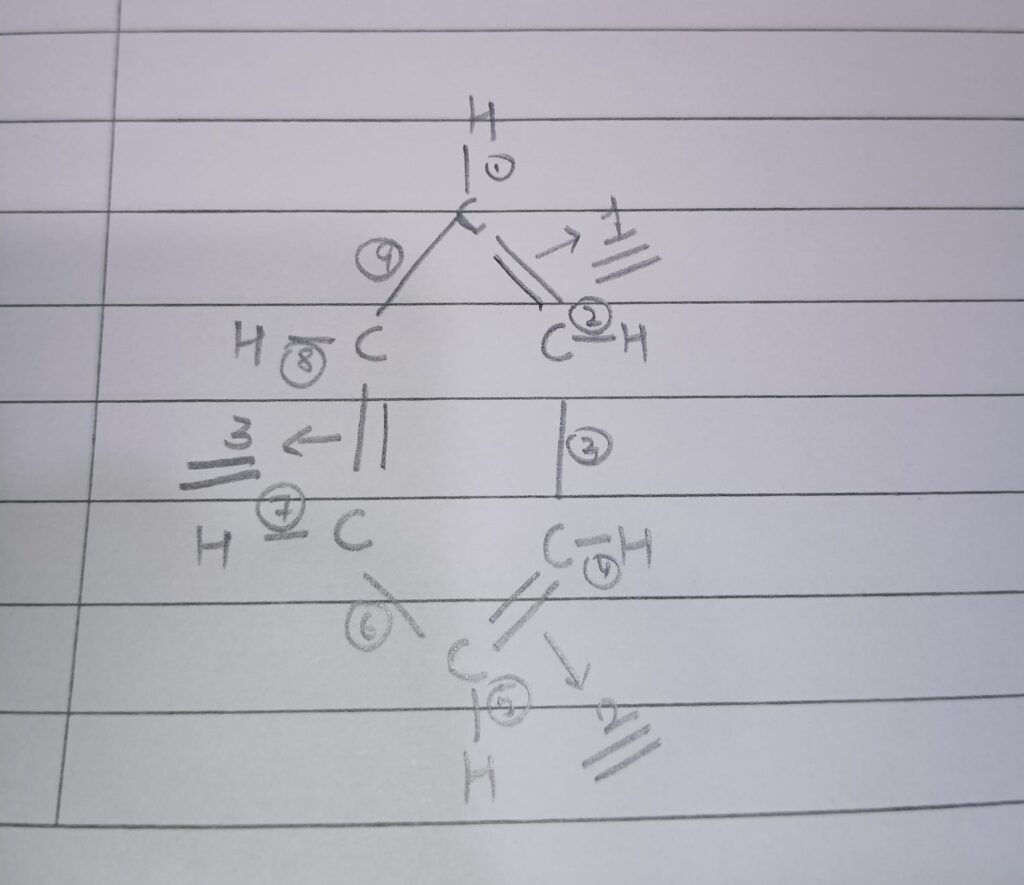
We can see in the structure of benzene that there are 6 C-H single bonds and 3 C-C single bonds. Also, there are 3 C-C double bonds. Hence, no of single bonds- 9 and no of double bonds- 3.
Ques 24. Carbon forms compounds mainly by covalent bonding. Why?
or
Carbon prefers to share its electrons rather than gaining or losing its electrons in order to attain noble gas configuration. Give reason to justify this statement.
Ans. Carbon mainly forms covalent bonds because it takes a lot of energy to form C4+ cation and C4- anion is very unstable in nature. And hence, it shares its electrons to form a covalent bond.
Ques 25. Define allotropes and hence write the name of allotropes of carbon.
Ans. Allotropes of an chemical element refers to its one or more physical forms that occur in same physical state but may have different physical and chemical properties.
Allotropes of carbon are – Diamond, Graphite and Buckminsterfullerene.
Ques 26. How can diamonds be synthesized?
Ans. Diamonds can be synthesised by subjecting pure carbon to very high pressure and temperature.
Ques 27. What are saturated and unsaturated carbon compounds?
Ans. Saturated hydrocarbons are those which contain only single bonds between carbon atoms, while unsaturated hydrocarbons have at least one double or triple bond between carbon atoms.
Ques 28. Why does substances burn with or without a flame?
Ans. Flame produces when a gaseous substance burn.For eg. When we ignite wood or charcoal, the volatile substances present
vapourise and burn with a flame in the beginning.when the substances do not vapourise or are non volatile they do not burn with a flame.
Ques 29. Write the oxidation reaction of ethanol.
Ans. When we oxidise alcohol, it converts into carboxylic acids.

Ques 30. What is denatured alcohol and why is alcohol used as a fuel?
Ans.To prevent the misuse of ethanol (alcohol) produced for industrial use, manufacturers add poisonous substances like methanol to make it unfit for drinking. They also add dyes to color the alcohol blue so that it can be identified easily. This form of alcohol is called denatured alcohol.
Alcohols are used as a fuel since it is a cleaner fuel which gives rise to only carbon dioxide and water on burning in sufficient air (oxygen).
3 marks Questions:
Ques 31. What happens when chlorine reats with methane in the presence of sunlight ? Write the name of reaction.
Ans. Chlorine replaces the hydrogen atom present in methane in the presence of sunlight. It is called a substitution reaction because one type of atom or a group of atoms takes the place of another.

Ques 32. Explain why carbon forms compounds mainly by covalent bonds. Explain in brief two main reasons for carbon forming a large number of compounds. Why does carbon form strong bonds with most other elements?
Ans. Carbon mainly forms covalent bond because it has 4 electrons and it is difficult to gain or lose 4 electrons to gain stability. That’s why sharing o of electrons happen and a covalent bond forms.
The two main reasons for carbon forming a large number of compounds are:
(i) Catenation i.e the self linking property of carbon to form a large number of compounds.
(ii) Tetravalency i.e its capacity to form 4 covalent bonds with carbon or any other element like hydrogen or oxygen etc.
Carbon forms strong bonds because of its smaller size that helps the nucleus to bind the shared pair of electrons easily.
Ques 33. (i) Draw the structural isomers of butane.
(ii) Draw the structures of propanol and propanone.
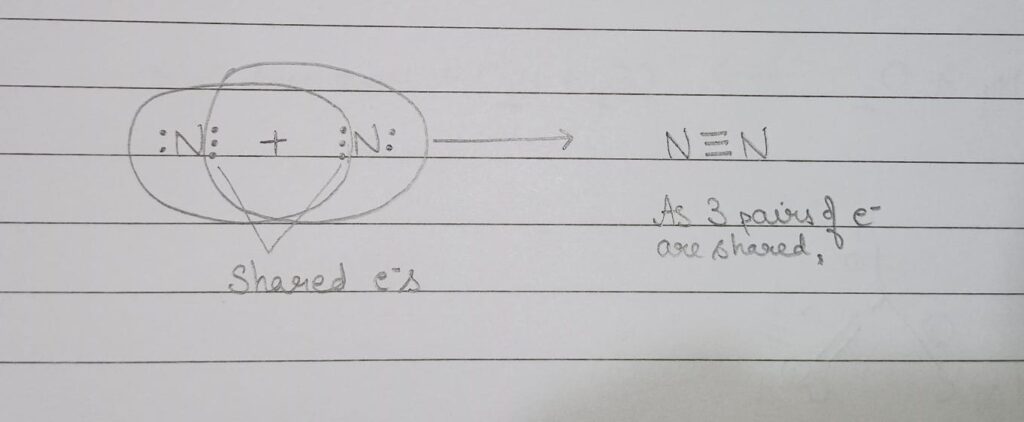
(iii) Show the covalent bond formation in Nitrogen molecule.
Ans. (i) Butane has following 2 structural isomers:

(ii) Structures of propanol and propanone are as follows:

(iii) Covalent bond formation in Nitrogen molecule:
As we know the atomic number of nitrogen is 7. so, its electronic configuration will be 2,5. We can see it has 5 valence electrons and need 3 electrons to complete its octet. To complete its octet nitrogen shares its 3 electrons as shown in image given below and forms a triple bond.
Ques 34. Define the following terms giving one example of each: (i) Alkynes (ii) Esterification (iii) Addition reaction
Ans. (i) Alkynes: Alkynes are the unsaturated hydrocarbons (having carbon-carbon triple bond) with the general formula CnH2n-2. Eg. C2H2
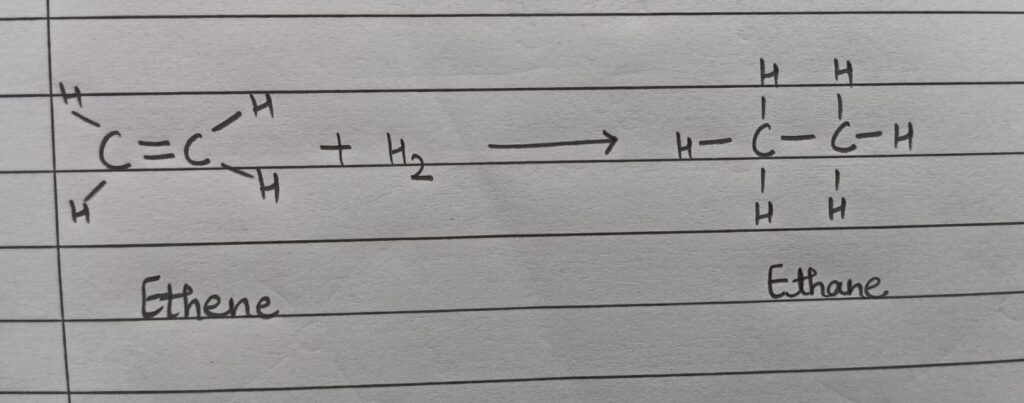
(ii) Esterification: Ethanoic acid reacts with absolute ethanol in the presence of an acid catalyst to give an ester –

This reaction between alcohol and carboxylic acid is known as esterification reaction.
(iii) Addition reaction: When an unsaturated hydrocarbon combines with another substance to form a saturated compound in the presence of a catalyst ( Ni/Pd) , the reaction is known as addition reaction. For eg.
This reaction is commonly used in the hydrogenation of vegetable oils using a nickel catalyst. Vegetable oils generally have long unsaturated carbon chains while animal fats have saturated carbon chains.
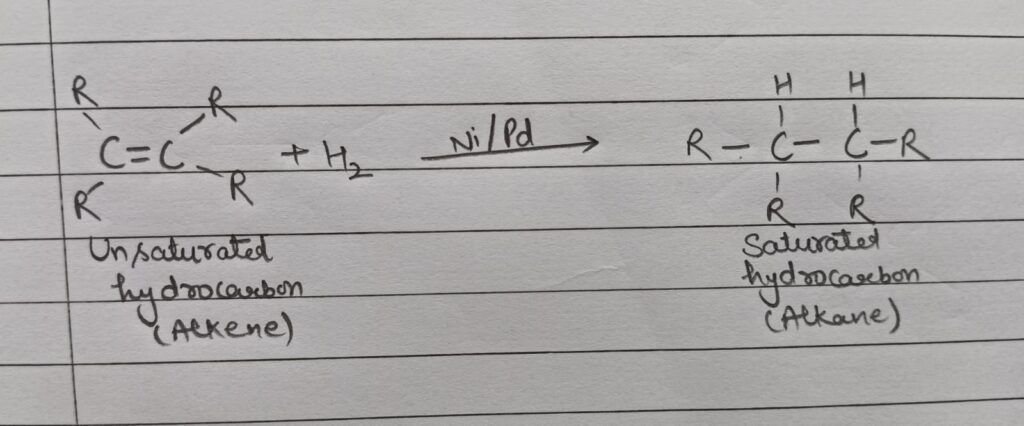
Here, Oils containing unsaturated fatty acids converts to saturated fatty acids.
Ques 35.Define catenation and oxidising agents.
Ans. Catenation: A carbon atom shows the self-linking property to form a large number of compounds which we call catenation.
Oxidising agents: Some substances are capable of adding oxygen to others. These substances are known as oxidising agents. for eg. Alkaline potassium permanganate or acidified potassium dichromate are oxidising alcohols to acids, that is, adding oxygen to the starting material.
These are some important questions and answers of carbon and its compounds class 10
To practice more, you can also check the NCERT Class 10 Science textbook from
https://ncert.nic.in/textbook.php
→ (Link to the NCERT Class 10 Science book)
https://cbseacademic.nic.in/
→ (For CBSE sample papers or syllabus)
Also checkout the notes of how do organisms reproduce
